Circio Holding ASA (”Circio” or ”the Company”) announced, on the 29th of April 2026, a research collaboration with Universidad de Santiago de Compostela (”USC”), through USC’s TraffikGene project, to evaluate non-viral delivery of circVec using TraffikGene’s proprietary peptide amphiphile carrier system. The aim is to assess whether the TraffikGene technology can deliver circVec into novel tissues for gene and cell therapy applications. The collaboration will proceed in three stages: in vitro screening of peptide carriers, physicochemical optimization of lead formulations, and in vivo evaluation in mouse models. TraffikGene is led by Prof. Javier Montenegro at CiQUS and is currently being spun out into an independent company. No financial terms were disclosed.
Analyst Group’s View on the TraffikGene Collaboration
For circVec to reach its full potential, Circio needs delivery technologies capable of bringing the circVec construct into the right cells, in the right tissues, safely and efficiently. circVec contributes the intracellular RNA stability advantage, but it relies on a delivery vehicle to physically transport the genetic payload to its target.
The TraffikGene collaboration adds a third such delivery engagement to Circio’s 2026 portfolio, following the in vivo CAR-M research collaboration with United Immunity in March and the technology evaluation agreement with Acuitas Therapeutics for in vivo CAR-T in April. Structurally, the TraffikGene agreement resembles the United Immunity setup: a joint research effort where Circio and the partner co-test the combined technology. Thus, distinct from Acuitas, which is structured as a technology evaluation that could potentially open an in-licensing pathway for Circio to access Acuitas’ clinically validated LNP platform.
What makes the TraffikGene engagement particularly interesting is that it introduces a delivery chemistry distinct from those evaluated to date. While United Immunity and Acuitas both rely on LNP-based systems for immune-cell targeting, TraffikGene uses peptide amphiphile carriers, which operate on a different physicochemical basis than LNPs. Analyst Group considers this the most material aspect of the announcement, as it reinforces Circio’s positioning of circVec as a delivery-agnostic expression platform, meaning circVec is, in theory, compatible with a wide range of delivery technologies rather than tied to any single carrier system. For the licensing thesis, this matters, as the broader the set of delivery formats circVec can plug into, the larger the universe of potential pharmaceutical partners.
A second notable element is the framing toward ”novel tissues” for both gene and cell therapy. Whereas United Immunity targets myeloid cells and Acuitas focuses on T-cells, both within the in vivo CAR thesis, TraffikGene’s stated ambition extends beyond immune-cell engineering. Non-viral delivery into tissues currently constrained by AAV’s limitations, payload size, immunogenicity, and the inability to redose. This represents a potentially meaningful broadening of circVec’s addressable applications, though the program remains at an early technical stage and tissue targets are still to be defined through the screening phase.
The agreement is also consistent with Circio’s stated objective, communicated at the Q1-26 update, of entering two to three new R&D technology collaborations on circVec AAV and in vivo CAR programs during H1-26. With three such collaborations now announced within approx. five weeks, Circio has already reached the upper end of that range with approx. two months remaining in H1-26. As with the prior 2026 collaborations, no financial terms were disclosed, which Analyst Group continues to view as a structural feature of these early-stage partnerships. Below is an updated overview of Circio’s disclosed external collaborations:
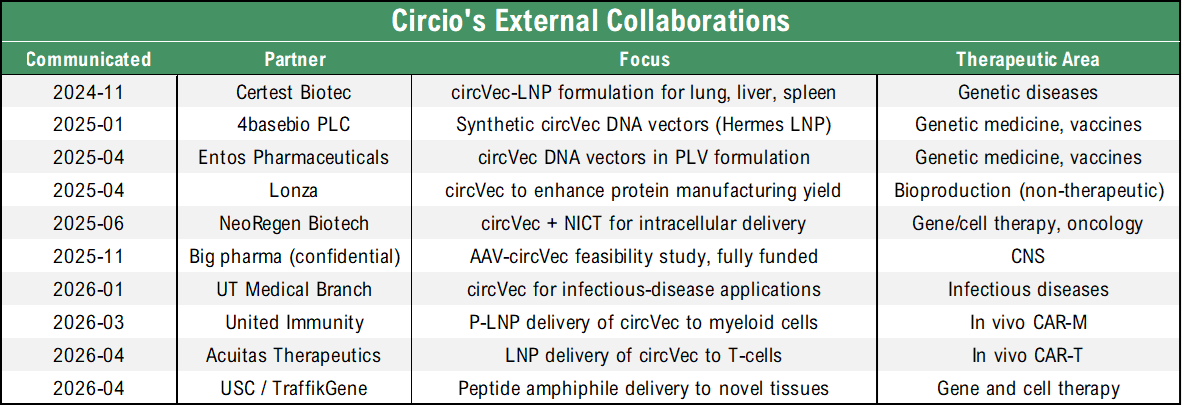
In summary, Analyst Group views the TraffikGene collaboration as a strategically coherent extension of Circio’s delivery partnership network that reinforces the delivery-agnostic positioning of circVec and broadens its potential reach beyond immune-cell applications. That said, the collaboration remains at a preclinical technical-feasibility stage with no disclosed financial terms, and the principal near-term value drivers continue to be the in vivo CAR-T data package expected during Q3/Q4-26, the disease-model efficacy readouts in eye and heart in Q4-26, and the partnered project update from the big pharma CNS feasibility study in Q4-26.